CAMBRIDGE, Mass., Jan. 13, 2025 (GLOBE NEWSWIRE) -- Immuneering Corporation (Nasdaq: IMRX), a clinical-stage oncology company seeking to develop and commercialize more effective and better tolerated therapies for cancer patients, today announced a positive update from its Phase 2a arm studying IMM-1-104 in combination with modified FOLFIRINOX (mFFX) in first-line pancreatic cancer, and provided a corporate update.
“We are thrilled to report two more responses in our Phase 2a arm studying IMM-1-104 in combination with modified FOLFIRINOX in first-line pancreatic cancer,” said Ben Zeskind, Ph.D., CEO of Immuneering. “The response rates emerging from both of our Phase 2a combination arms in first-line pancreatic cancer are comparable to one another, and speak not only to IMM-1-104’s potential to drive a new standard of care in pancreatic cancer, but also its potential as a first-of-its-kind, well-tolerated MEK inhibitor that could be safely used in a variety of combinations to drive better outcomes for patients across a range of indications.”
Zeskind continued: “Building on our positive January 7, 2025 data update, we strengthened our cash balance with nearly $14 million in net proceeds raised through our ATM facility. We began 2025 strong and intend to sustain that momentum throughout the year, including with a planned second quarter 2025 Phase 2a trial data update, the anticipated initiation of new combination trial arms, and planning for a pivotal trial of IMM-1-104 in combination with modified gemcitabine/nab-paclitaxel.”
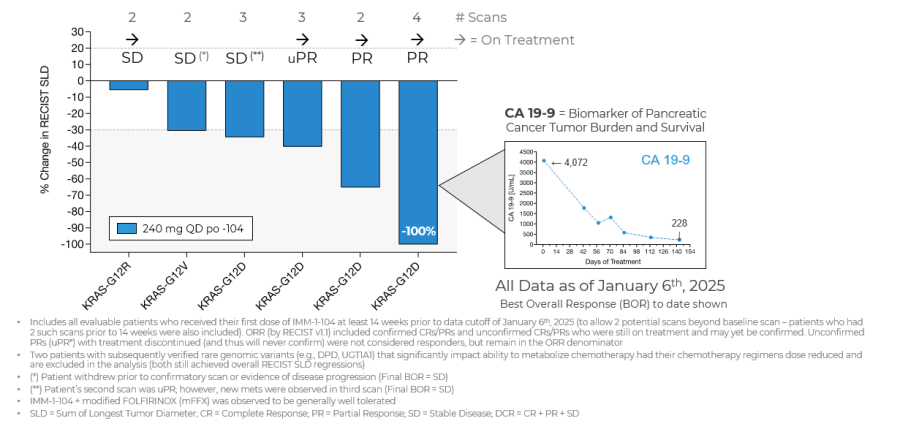
Data Update from Phase 2a Arm Evaluating IMM-1-104 with Modified FOLFIRINOX in First Line Pancreatic Cancer as of January 6, 2025
Source: Immuneering Corporation
Additionally, Immuneering today announced that it raised net proceeds of $13.7 million through utilization of its ATM facility following its January 7, 2025 update from its ongoing Phase 2a clinical trial.
Near-Term Milestone Expectations
IMM-1-104
About Immuneering Corporation
Immuneering is a clinical-stage oncology company seeking to develop and commercialize more effective and better tolerated therapies for cancer patients. The Company’s lead product candidate, IMM-1-104, is an oral, once-daily deep cyclic inhibitor of MEK designed to improve tolerability and expand indications to include RAS-driven tumors such as most pancreatic cancers. IMM-1-104 is currently in a Phase 1/2a trial in patients with advanced solid tumors including pancreatic cancer. IMM-6-415 is an oral, twice-daily deep cyclic inhibitor of MEK currently in a Phase 1/2a trial in patients with advanced solid tumors harboring RAS or RAF mutations. The company’s development pipeline also includes several early-stage programs. For more information, please visit www.immuneering.com.
Forward-Looking Statements
This press release contains forward-looking statements, including within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including, without limitation, statements regarding: Immuneering’s plans to develop, manufacture and commercialize its product candidates; the treatment potential of IMM-1-104, alone or in combination with other agents, including chemotherapy, checkpoint inhibitors and BRAF inhibitors; the plans and objectives of Company management for future operations, including with respect to the planning and execution of additional IMM-1-104 combination trials and potential pivotal trial of IMM-1-104 in combination with modified gemcitabine/nab-paclitaxel; and the timing for release of additional results from the Phase 2a portion of the trial for IMM-1-104.
These forward-looking statements are based on management’s current expectations. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: the risks inherent in oncology drug research and development, including target discovery, target validation, lead compound identification, and lead compound optimization; we have incurred significant losses, are not currently profitable and may never become profitable; our projected cash runway; our need for additional funding and ability to continue as a going concern; our unproven approach to therapeutic intervention; our ability to address regulatory questions and the uncertainties relating to regulatory filings, reviews and approvals; the lengthy, expensive, and uncertain process of clinical drug development, including potential delays in or failure to obtain regulatory approvals; our reliance on third parties and collaborators to conduct our clinical trials, manufacture our product candidates, and develop and commercialize our product candidates, if approved; failure to compete successfully against other drug companies; protection of our proprietary technology and the confidentiality of our trade secrets; potential lawsuits for, or claims of, infringement of third-party intellectual property or challenges to the ownership of our intellectual property; our patents being found invalid or unenforceable; costs and resources of operating as a public company; and unfavorable or no analyst research or reports.
These and other important factors discussed under the caption “Risk Factors” in our Quarterly Report on Form 10-Q for the period ended September 30, 2024, and our other reports filed with the U.S. Securities and Exchange Commission, could cause actual results to differ materially from those indicated by the forward-looking statements made in this press release. Any such forward-looking statements represent management's estimates as of the date of this press release. While we may elect to update such forward-looking statements at some point in the future, except as required by law, we disclaim any obligation to do so, even if subsequent events cause our views to change. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this press release.
Media Contact:
Gina Nugent
This email address is being protected from spambots. You need JavaScript enabled to view it.
Investor Contact:
Laurence Watts
619-916-7620
This email address is being protected from spambots. You need JavaScript enabled to view it.

| Last Trade: | US$1.95 |
| Daily Change: | -0.06 -2.99 |
| Daily Volume: | 1,923,784 |
| Market Cap: | US$60.550M |
December 19, 2024 November 13, 2024 October 15, 2024 | |

Hillcrest Energy Technologies is a clean technology company developing high value, high performance power conversion technologies and digital control systems for next-generation powertrains and grid-connected renewable...
CLICK TO LEARN MORE
Leveraging its vertically-integrated approach from mine to material manufacturing, Graphite One intends to produce high-grade anode material for the lithium-ion electric vehicle battery market and energy storage systems...
CLICK TO LEARN MORECOPYRIGHT ©2022 GREEN STOCK NEWS